HIGHER YIELDS
KNO3 increases marketable yields of larger-sized fruit with intense color, both applied as foliar spray or in fertigation.
STRONGER TREES
KNO3 improves tree health and boosts tree’s defenses like HLB (citrus greening)
Samples are taken from mature leaves (4-7 months) on a non-fruit-bearing spring shoot (the fifth to seventh leaffrom the apex). Sampling takes place in February and March.
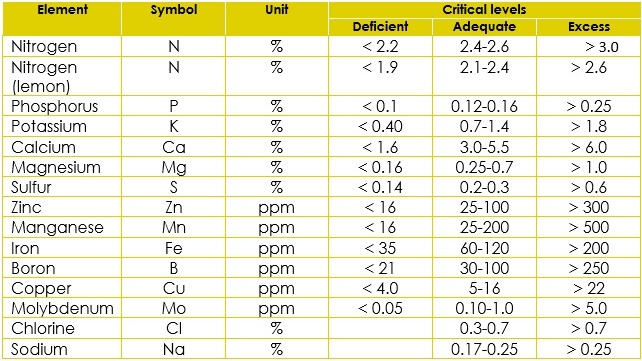
The leaf symptoms of deficiencies in these species are very similar, and deficiencies in more than one element are also common. Consequently, anomalous development isidentified only via leaf analysis (Table 6). Table 6 Critical levels for citrus (dry weight basis)
Table 6 Critical levels for citrus (dry weight basis)
Source: Image of citrus from SQM image library
In orange trees, excess nitrogen (N) has serious negative impact, delayingripening and diminishing fruit quality. The Mitscherlich yield response curveapplies nitrogen (N) dosage in citrus.
Deficiency
Nitrogen (N) is more important than other nutrients and greatly influences development and yield. Too little nitrogen (N) results in generalized chlorosis and impacts growth,especially in new plantings. Nitrogen (N) is particularly important for flowering. The greatest nitrogen (N) expenditure is in the fruit, flowers and leaves, even though they contain equal amounts of potassium (K) andcalcium (Ca). Plants withhigh or "luxury” nitrogen (N) levels have been observed to be more frost-resistant than thosewith low levels. Unlike other fruit species, oranges can be fertilized with nitrogen (N) atany time of the year. Their roots absorb nitrogen (N) quickly. Since the leaves are asignificant nitrogen (N) reserve, new plantations can have enough nitrogen (N)stores to last a few months, even in nitrogen-scarce soil. Excess Nitrogen (N) High doses of nitrogen (N) can cause one or some of the following detrimental effectson fruit quality:
Deficiency Correction
Nitrate deficiency is corrected by applying a nitrogen (N) carrier. Citrus can be fertilizedwith nitrogen (N) at any time of the year, as long as it is applied at the same time of yearand there is sufficient availability at flowering. Various nitrogen (N) carrier types have been used successfully, including urea, ammonium sulfateand nitrate.
Deficiency
Phosphorus (P) is bound to essential components like enzymes and others that participate innormal cell function. Phosphorus deficiency is uncommon in citrus and does not affect fruit size. A phosphorus deficiency has the following impacts:
Deficiency Correction
Phosphorus deficiency is corrected by applying a granulated phosphorous (P) carrier to the soil. Increasing the leaf content has not been shown to produce toxicity.
Deficiency
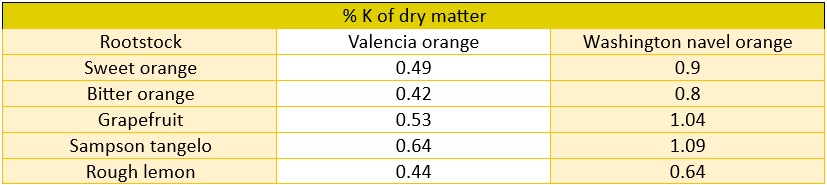
Despite the large quantities in plants, potassium (K) is not amongthe established biochemical compounds, and its functions are not entirely clear. It is an excellent cation,neutralizing the acid radicals constantly produced by the plants’ metabolism. Potassium (K) is a cation that creates internal osmotic potential, enablingwater to enter the vacuole and the cell to grow. The cell wall musthave great capacity to expand and accumulate solutes. Potassium (K) maintains an internal water balance,regulating the opening and closing of stomata.Potassium (K) is presumed to have a role in metabolic processes like cell division innew tissues. One clear symptom of potassium (K) deficiency is in the terminal leaves, which take ona curled shape. Severe deficiency stunts growth and causes interveinal chlorosisand twig dieback. Potassium (K) deficiency is frequently associated withiron (Fe) deficiency. The main symptoms of potassium (K) deficiency are smaller fruit size and prematurefruit drop. Low potassium (K) content affects grapefruit yield. Adequatepotassium (K) content considerably improves post-harvest fruit conservation.Given the very wide range between low and adequate content, normal vegetative growth is unaffected. Different rootstocks have varying potassium (K) absorption abilities.In decreasing order, grafts of Valencia orange trees with grapefruit, sweet orange and bitter orange rootstocks tend toincrease the potassium (K) content in leaves compared to the rough lemon,Cleopatra mandarin and Rusk citrange, which had the lowest potassium (K) levels (Reuther andSmith, 1950). The differentiated absorption of the same orange tree species on different rootstocksis shown on Table 7.
Table 7 The effect of rootstock on potassium (K) composition in two different varieties, according to Chapman and Brown as cited in (5).
Increasing potassium (K) levels produces the following effects:
Deficiency Correction
Potassium (K) deficiency is corrected by applying a potassium (K) carrier to the soil. Potassium sulfate (K2SO4)or potassium nitrate (KNO3) is preferable to potassium chloride (KCl) due to the susceptibility of the latter toexcess chloride. For automatic irrigation, use the same products in theirsoluble formulations.
Calcium (Ca) pectate is a cementing substance that binds cells together. Calcium(Ca) neutralizes or precipitates certain organic acids produced in the plant’s metabolic activities. It precipitates oxalic acid; acts as an antidote to the toxicities ofother bases like sodium (Na), potassium (K) and magnesium (Mg); and is important for the development and function ofthe root system, which is damaged by deficiency. Calcium (Ca) is the element with the highest content in citrus fruit. Given its very low mobility,Calcium (Ca) content is almost invariable over the leaf’s life. There is competition/antagonism between calcium (Ca) and nitrogen (N) and, to a very minor extent, with potassium (K).
Deficiency
Calcium (Ca) deficiency is rare in Chile since it occurs in soils with acid pH andexcess potassium (K), a highly improbable situation in the areas and soils wherecitrus grows.
Deficiency Correction
Apply agricultural gypsum or calcium sulfate dihydrate (CaSO4 * 2H2O) toincrease calcium (Ca) in the soil’s cation exchange. Depending on the irrigation system, granular or soluble calcium nitrate can be included in the fertilization schedule.
Deficiency
Magnesium (Mg) activates more than 12 enzymes associated with the metabolism ofcarbohydrates. Magnesium (Mg) is a component of chlorophyll; consequently, too little produceschlorosis. In citrus, deficiency symptoms appear in mature leaves as"bronzing," defoliation, frost susceptibility, poor root development and twig dieback. In addition, yields fall and the deficiency leads to "alternate" production and poor fruit quality. Symptoms occur most frequently in late summer and autumn. Yellowish necrotic spots appear in the interveinal area. Unlike nitrogen (N) deficiency, the midrib retains its colorlonger. Trees with magnesium (Mg)deficiency are more susceptible to frost damage. For unclear reasons, magnesium (Mg) has a synergistic effect on zinc (Zn) andmanganese (Mn), increasing the leaf content of these elements. A Ca:Mg ratio around 7:1 is adequate. Ten or more indicatesa deficiency.
Deficiency Correction
Magnesium (Mg)deficiency is corrected with magnesium sulfate, also known as Epsom salt (MgSO4), applied pre-planting or via topdressing. Spraying Epsom salt has not been shown to produce good results. Alternatively, magnesium nitrate (MgNO3) can be sprayed at 1% - 1.5%.
Deficiency
Sulfur (S) is a component of several amino acids, proteins and vitamins. Sulfur-deficient new leaveshave less than 0.1% (b.p.s), and toxicity occurs at about1% (b.p.s). Leaves may have slight chlorosis, and vitamin C content is low.
Deficiency Correction
Sulfur (S) deficiency is corrected with magnesium sulfate, also known as Epsom salt (MgSO4), potassium sulfate(K2SO4) and/or adding sulfuric acid to the automatic irrigation system.
Deficiency
Zinc (Zn) deficiency is among the most common deficiencies in citrus. Among other functions, zinc (Zn) is aprecursor to growth hormones like auxins, as it is involved in the formation of theamino acid tryptophan. Zinc (Zn) activates enzymes in protein formation and promotesproper root development. It is important for twig and leaf growthas well as fruit formation. A zinc (Zn) deficiency manifests as chlorosis, particularly in new leaves on terminal shoots; “little leaf" (small, abnormally narrow leaves) or shoot dieback. Leaf mottling (foliocellosis) occurs. Chlorophyll disappears from the interveinal zone. Orange trees are the most susceptible citrus variety to this type of chlorosis. The fruit is smaller andpale in color. When the deficiency is severe, there is twig dieback on the plant’s interior. Since copper (Cu) and zinc (Zn) have antagonistic effects, their application should be spaced out. As with other minor elements, excess is more hazardous than deficiency.
Deficiency Correction
Zinc (Zn) deficiency is corrected by spraying during autumn bud break and spring flush when tender leavesare more absorbent. Zinc sulfate (ZnSO4) at 0.3%(300 gr/100 lt of water), neutralizing with 30 cc/100 lt of concentrated sodiumhydroxide (NaOH) solution, can be sprayed. Alternatively, commercial spray productswith high zinc (Zn) concentrations can also be used. Since zinc (Zn) availability is pH dependent, applications, e.g., zinc sulfates, to high-pH soilare inefficient. Given that more acidic soil increases the element’s availability, adding sulfuric acid (H2SO4)to the automatic irrigation system has yielded good results. Zinc (Zn) application is recommended post-flowering when symptoms appear.
Deficiency
Manganese (Mn) reduces Fe3+ to Fe2+ in the plant, aiding inchlorophyll synthesis and nitrate assimilation. It is also essential to CO2 assimilation inphotosynthesis, acting in the formation of riboflavin, ascorbic acid and carotene. Deficiency produces discoloration (yellowing), very typically in the interveinal area. Certain soil conditions may lead to manganese (Mn) deficiency, the most common being:
Deficiency Correction
Leaf applications are very effective in correcting manganese (Mn) deficiency. Manganese sulfate (MnSO4) at 0.35% (350 g/100 lt of water), neutralizing with 30 cc/100 lt ofof concentrated caustic soda solution (NaOH, prepared with 1 kg NaOH in 1 ltwater) can be used. If the soil pH is below 6.0, soil application has proven efficient (Florida,USA). Alternatively, the soil can be made more acidic by adding sulfuric acid (H2SO4) tothe automatic irrigation system. When both zinc (Zn) and manganese (Mn) levels are deficient, zinc sulfate (ZnSO4)at 0.4% and manganese sulfate (MnSO4) at 0.3% can be used in a mixture, neutralized with aconcentrated caustic soda (NaOH) solution of 30 cc/100 lt of water.
Deficiency
Although it is not a constituent of chlorophyll, iron (Fe) is related to chlorophyll formation. Consequently,the chlorophyll content of tissues is related to soluble iron (Fe),which acts on enzymes that affect respiration and aids energy transfer. Iron (Fe) alsoparticipates in the formation of some proteins. Iron (Fe) deficiency produces chlorosis, though the ribbing remains green. Severe iron (Fe) deficiencyaffects plant development, and limb and twig dieback occur. Some conditions that increase susceptibility to iron (Fe) deficiency are:
Deficiency Correction
Iron (Fe) deficiency is corrected by acidifying the soil with sulfur (S), sulfuric acid (H2SO4) orammonium sulfate (NH4SO4), which are nitrogen (N) sources and acidifying agents. Sprays withamino acids containing iron (Fe) have been successful. Applications of iron chelates (Fe-EDTA, Fe-DTPA and Fe-EDDHA) and/or sulfuric acid (H2SO4)via an automated irrigation system are efficient control measures.
Deficiency
Boron (B), like other minor elements, plays a catalytic or enzymatic role incell wall synthesis (formation of the pectin and lignin in cell walls). Boron (B) is essential to maintaining a balance between sugars and starch, acting inthe translocation of sugar and carbohydrates. It is important in pollination, seed production,cell division, nitrogen (N) metabolism, protein formationand ribonucleic acid (RNA) synthesis. During flowering, boron (B) improves pollen germination,subsequent development of the pollen tube, fruit set and metabolismof phenols and ascorbates. Boron (B) is also required at points of high metabolic activity, likethe apices of shoots and roots, for the processes of cell division, cellular elongation and meristematic differentiation, according to Palma Boron (B) deficiency symptoms include:
Deficiency Correction
Boron (B) deficiency is corrected with spray or soil applications. Spraying withsodium octaborate (NaHBO3), with 44% B2O3 at a ratio of 1 kg/1000 lt of water, in the spring is one option. Another is applying 20-25 grams of borax per plant inside the raised ring around the trunk or with anyboron-based leaf fertilizer, e.g., Speedfol® B SP. Solubor sprays arealso recommended (1 kg/1000 lt. of water) for the tender autumn and spring shoots.
Deficiency
Copper (Cu) acts as a coenzyme in several enzymatic systems linked to theconversion of amino acids into proteins. Copper (Cu) influences chlorophyll formation. Likezinc (Zn), it acts as a catalyst and regulator in oxide-reduction reactions. Copper (Cu) deficiency causes chlorosis and twig dieback. It has not beendetected in Chile, where citrus trees are sprayed with copper-based products to control and/orprevent brown rot (P. citrophthora). Though it does not occur in Chile, copper (Cu) deficiency is common in organic soils. The fruit from copper-deficient trees has dark brown indentations, and the leaves are an intense green,giving an impression of excess nitrogen (N). The twigs curve in anS-shape, and the leaves have reddish-brown pustules.
Deficiency Correction
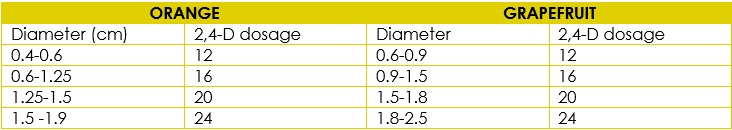
Copper (Cu) deficiency is corrected by spraying with copper sulfate (CuSO4) at 0.05% in the autumn or by sprayingBordeaux mixture or another copper (Cu) product. These treatments must be applied before the rainy season. Supplemental Hormonal Information - The Use of 2,4-D to Improve Fruit Size andMinimize the Drop2,4-D applied to orange trees improves fruit size. The dosage depends on thefruit diameter and species (Table 8).
Citrus plants are sensitive to chlorides (Cl) and/or sodium (Na). These components should ideally bein both soil and irrigation water, particularly in arid areas.
We will send you a link to your email, then you will follow the instructions that will appear to retrieve your password.
This website uses its own and third-party cookies to collect information in order to improve our services, to show you the advertising related to your preferences. If you continue browsing, it implies acceptance of their installation. The user has the possibility to configure his browser, if he wishes, to prevent them from being installed on his hard disk, although he must bear in mind that this possibility can be useful in navigating the web page.
For more information read here ACCEPT